Connect With a Global Community of Experts
As the only event laser-focused on raw materials for cell and gene therapy, the 2nd Raw Materials for Cell & Gene Therapy Summit provided an unmatched platform for suppliers, CDMOs, CROs, and consultants to connect directly with decision-makers shaping the future of CGT manufacturing standards.
Unlike broader conferences, the Summit drilled deep into materials science, regulatory alignment, and supplier strategy, key factors influencing therapeutic success. With the FDA, USP, and leading biotechs like Ultragenyx, Vertex and Abeona in attendance alongside pharma leaders such as Bristol Myers Squibb and Roche, this was where the future of CGT quality took shape and raw materials took center stage, making it a premier partnership opportunity.

Why Partner with the Summit?
Establish Your Brand as a Raw Materials Thought Leader
Position your organization at the forefront of one of the most complex and rapidly evolving areas in CGT. Through speaking opportunities, panel involvement, and curated roundtables, you’ll elevate your visibility as a trusted partner in raw material innovation and compliance
Drive Qualified Pipeline Growth & Partnerships
This Summit is engineered for results, bringing together buyers and influencers with real purchasing power. Whether you're a supplier, CDMO, CRO, or consultant, you'll gain access to decision-makers ready to collaborate, co-develop, and source smarter for next-gen therapies
Access Focused Cell & Gene Therapy Raw Materials Audiences
Engage directly with decision-makers in MSAT, Quality, Supply Chain, and Regulatory Affairs who are actively shaping raw material strategies for clinical and commercial CGT pipelines. This is your opportunity to showcase solutions to biopharma stakeholders tasked with vendor qualification, testing, and risk mitigation
Hear What Our Past Sponsors Have to Say

Raw materials and material qualification is an emerging field and knowledge sharing is key in the overall success of the cell and gene therapy space. A lot of suppliers cannot support the needs of the cell and gene therapy raw material quality attributes and establishing a base line for everyone to be on the same page is the beginning
Sona Bairamian, Director, Materials Science, Atara Biotherapeutics
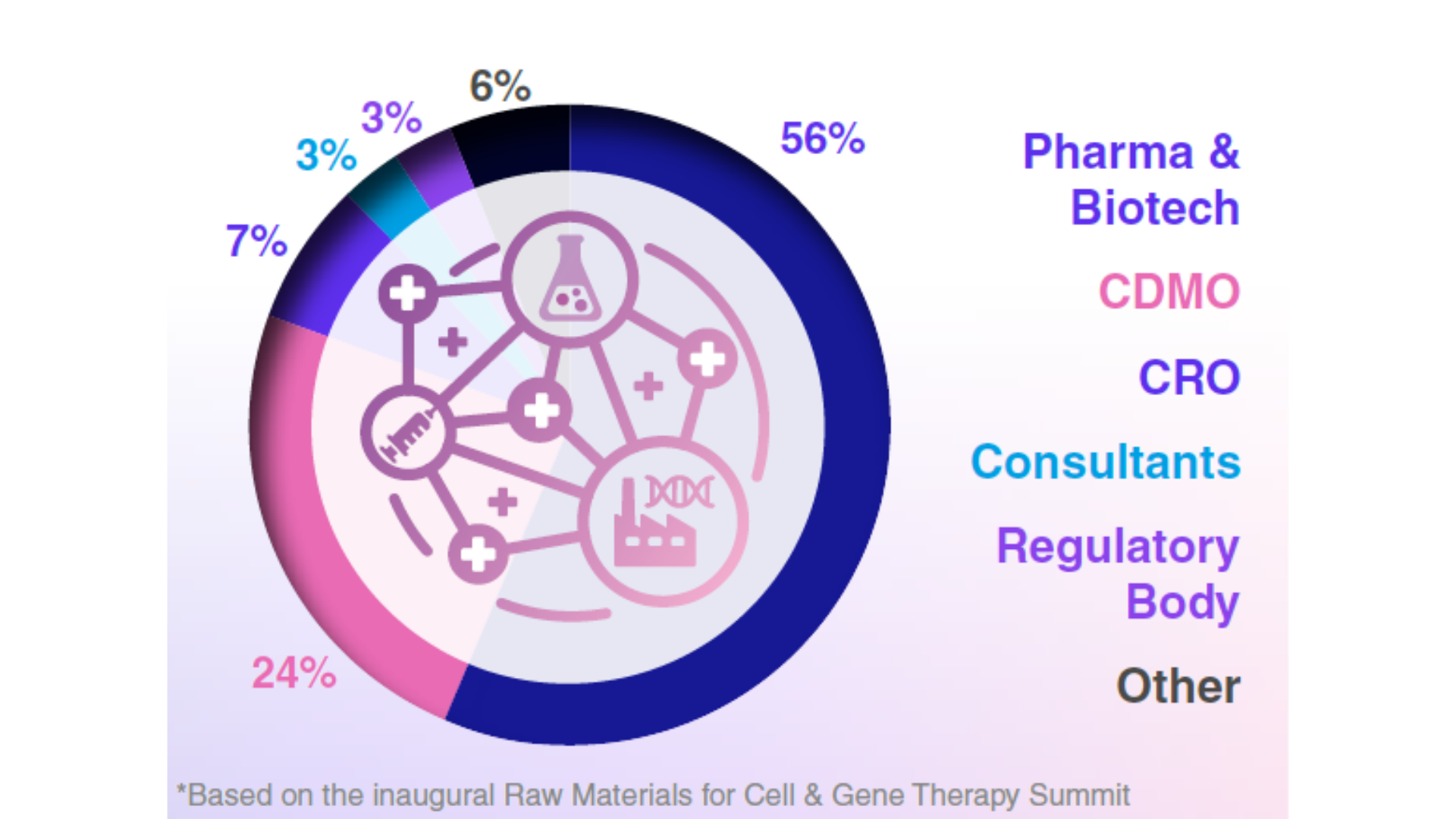
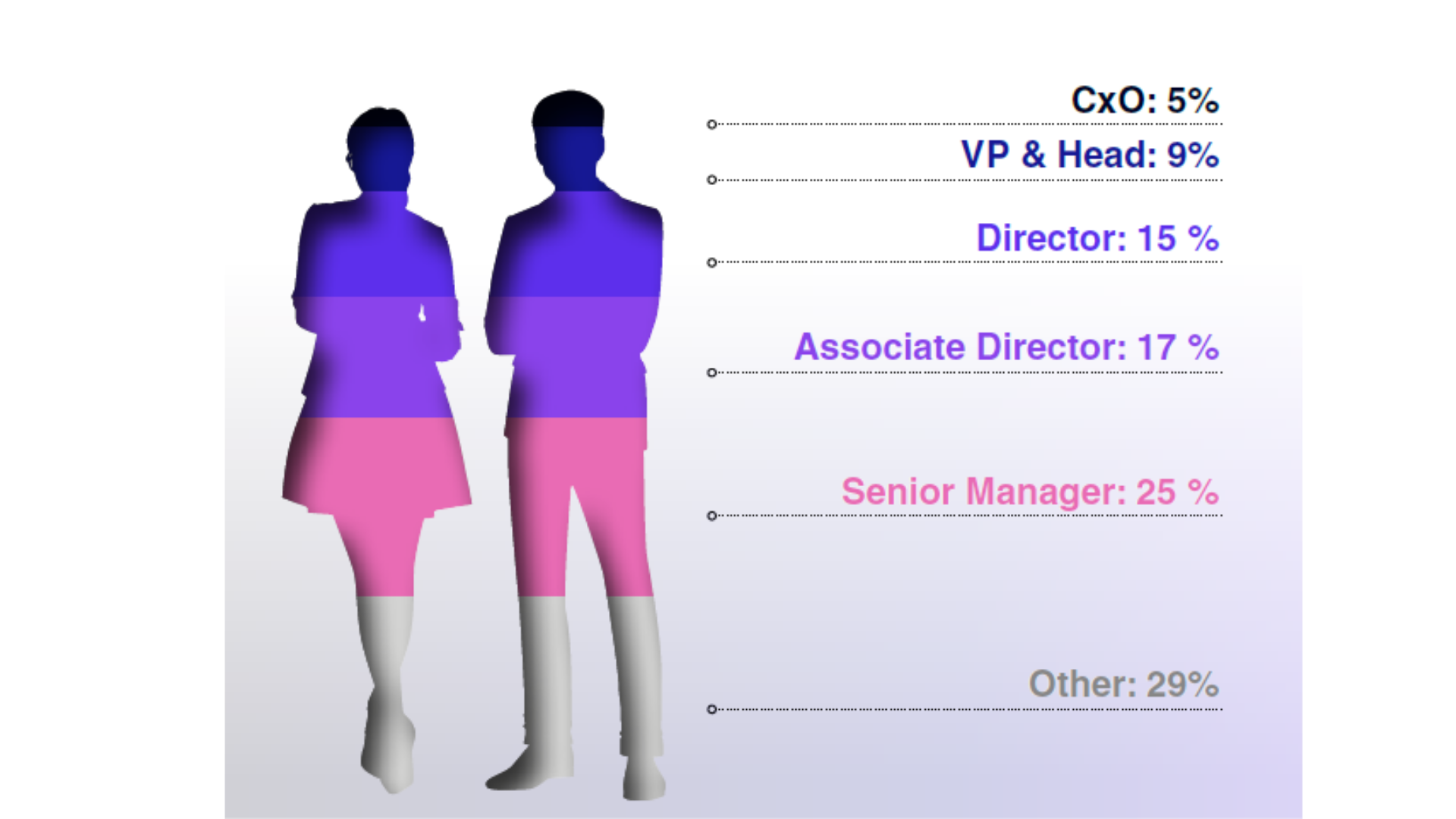
Audience Composition
Company Type
Attendee Seniority
Attending Companies Include









